This site is for Residents of Europe, Middle East, Africa, Latin America & Asia Pacific. If you are looking for career opportunities in Chinese Mainland, please click here.
![UltrawideMerckJogWebBanner[66].jpg-1760025804973](https://cdn.phenompeople.com/CareerConnectResources/MSD1GB/images/UltrawideMerckJogWebBanner[66].jpg-1760025804973-1762257955243.jpg)
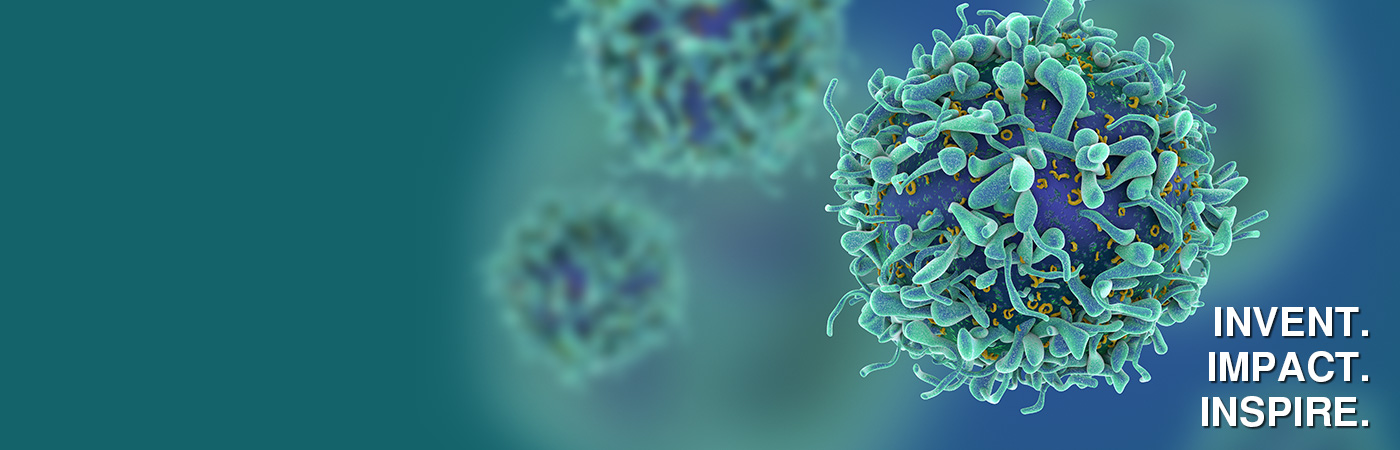
Together, we can make an impact.
Your journey starts here - driven by purpose, powered by people.
Find your fit!
Your curiosity can help change lives.
Join a team turning innovation into breakthroughs that help improve the health of people and animals across the world. We invest in your growth, wellbeing and future — because when you thrive, so does our purpose.
Explore our divisions where your impact can make a real difference.
Explore our divisions where your impact can make a real difference.

Meta card 4
MSD Manufacturing Division
Join the teams contributing to saving and improving lives through medicines and vaccines that help people and animals worldwide.
Learn more >
Learn more >

Meta card 1
Global Support Functions
Bring your expertise to a global team united by one purpose — to help save and improve lives.
Learn more >
Learn more >

Meta card 4
MSD Research Labs
Join the teams where science meets purpose and connect curiosity with compassion — using innovation to tackle the world’s toughest health challenges.
Learn more >
Learn more >

Meta card 2
Human Health
Join the teams that bring our science to life for patients around the world.
In human health, you’ll assist in connecting innovation with the people who need it most.
Learn more >
In human health, you’ll assist in connecting innovation with the people who need it most.
Learn more >

Meta card 3
Animal Health
Join a global team dedicated to advancing animal health and wellbeing, working together to protect animals, support customers and strengthen the world’s food supply.
Learn more >
Learn more >
Your engaging footer subtitle goes here.
As a leader in biopharmaceuticals, we focus on scientific innovation to deliver medicines and vaccines that may help millions of people around the world.

Bring your talent and expertise to teams helping to save and improve lives.
Learn more >

Use your skills and ambition to grow your career with a team that invests in you.
Learn more >

Be yourself, grow with purpose and help build a culture where everyone feels included.
Learn more >

Inclusion and belonging
Here, you’re empowered to bring your authentic self and ideas that spark innovation.
Learn more >
Discover what life at MSD is all about.
Explore our culture, benefits and how we support your wellbeing and career journey.
Compensation and benefits
Explore how we support your financial wellbeing, family and future in and outside of work.
Learn more >
Learn more >
Culture
Discover what it’s like to work in an environment where collaboration, respect and curiosity thrive.
Learn more >
Learn more >
Health and wellbeing
Your health. Your balance. Your future.
Learn how we create a workplace that supports your physical, mental and emotional wellbeing every day.
Learn more >
Learn how we create a workplace that supports your physical, mental and emotional wellbeing every day.
Learn more >
How we hire
Your journey starts here.
Understand our hiring process and get tips to help you find the role that fits your skills and aspirations.
Learn more >
Understand our hiring process and get tips to help you find the role that fits your skills and aspirations.
Learn more >
Your engaging footer subtitle goes here.

Join our talent community today.
Receive updates on career opportunities tailored just for you.
Connect with us on social!.
Our Awards
Our Awards






This careers website is intended for Residents of Europe, Middle East, Africa, Latin America and Asia Pacific. Information on this site is for the purpose of presenting career opportunities at our organisation and giving our audience a view of our culture, diversity and growth opportunities. Our Company is an equal opportunity employer, proudly embracing diversity in all of its manifestations.